How to Reconstitute Peptides: Safe Mixing Instructions
How to Reconstitute Peptides: Safe Mixing Instructions
Reconstituting peptides involves dissolving freeze-dried peptide powder into a liquid solution for accurate dosing and research use. This process requires precision and sterility to maintain peptide integrity and avoid contamination. Here's a quick breakdown of the essentials:
- Preparation: Use bacteriostatic water (preferred for multi-dose vials) or sterile water (for single-use). Gather sterile syringes, alcohol swabs, and nitrile gloves as part of a sterile technique.
- Mixing: Inject diluent slowly against the vial wall to avoid damaging the peptide. Never shake - gently swirl to dissolve.
- Storage: Refrigerate the solution at 36–46°F (2–8°C) and use within 28–30 days (if bacteriostatic water is used).
- Dosage: Calculate concentrations carefully to ensure dosing accuracy.
Proper handling ensures the solution remains stable and effective for your research. Avoid freezing, and discard solutions showing signs of contamination, discoloration, or cloudiness.
Reconstituting Your Peptides - Self Administration (A Tutorial)
sbb-itb-7fad836
Tools and Materials You'll Need
To ensure a sterile and effective reconstitution process, gather all the necessary supplies beforehand. You’ll need a peptide vial containing lyophilized (freeze-dried) peptide powder, bacteriostatic water (a common diluent containing 0.9% benzyl alcohol), and insulin syringes in sizes like 1 mL (100 units), 0.5 mL (50 units), or 0.3 mL (30 units). Use 70% isopropyl alcohol swabs to sterilize vial stoppers before inserting needles. Wear nitrile gloves for added protection and dispose of used needles in a sharps container. Proper preparation of your workspace and tools is essential for safe handling.
Basic Equipment
Start by disinfecting a clean, flat surface with isopropyl alcohol. Label each vial clearly with the peptide name, concentration, and the date of reconstitution immediately after mixing. Always use a fresh, sterile syringe for each draw. Before inserting a needle, sterilize the vial’s rubber stopper with an alcohol swab and allow it to air dry for 15–30 seconds.
Selecting Quality Peptides
Inspect your peptide vial carefully before use. The peptide should appear as a dry cake or loose powder. If you notice any signs of moisture, discoloration, or a yellow or brown tint, the peptide may be damaged and should not be used. To ensure the integrity of your materials, always source peptides from reliable suppliers that provide properly lyophilized products capable of maintaining structural stability during reconstitution and storage.
Types of Diluents
The choice of diluent plays a key role in maintaining peptide stability. Bacteriostatic water is the preferred option for multi-dose vials because its 0.9% benzyl alcohol content helps preserve the solution for up to 28 days when stored at 36–46°F (2–8°C). Sterile water, on the other hand, contains no preservatives and is suitable only for immediate, single-dose use, as it becomes susceptible to contamination after the first puncture.
For specialized peptides, such as IGF-1 LR3, 0.1M acetic acid may be required, while hydrophobic peptides might need DMSO for proper dissolution. Always allow the diluent to reach room temperature for 5–10 minutes before mixing. This step helps prevent clumping and ensures the peptide dissolves completely. Using the correct diluent is essential for maintaining accuracy in concentration and stability, which directly impacts proper dosing later in the process.
How to Reconstitute Peptides Step-by-Step
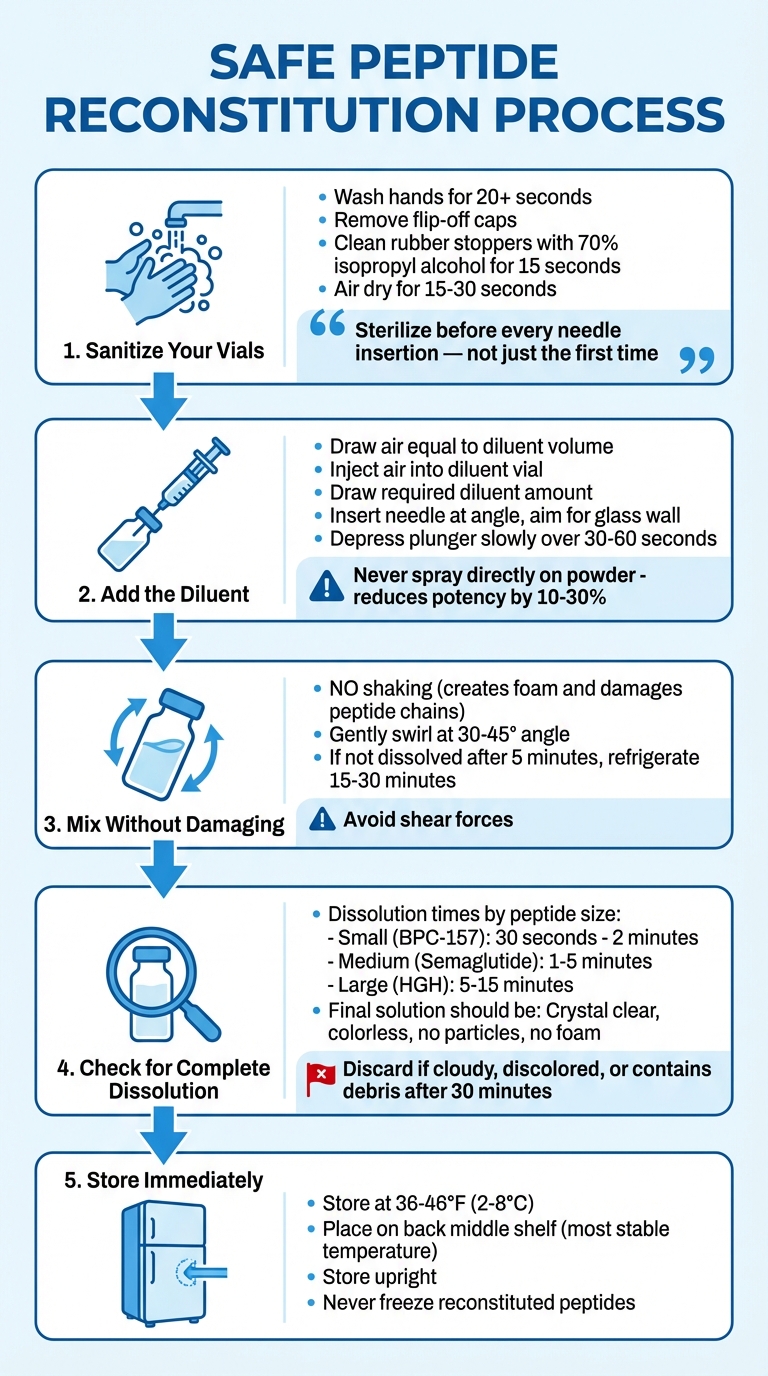
Step-by-Step Guide to Reconstituting Peptides Safely
When reconstituting peptides, precision and sterility are key. Every step requires careful handling to maintain the peptide's integrity and avoid contamination.
Sanitizing Your Vials
Start by washing your hands thoroughly with soap and water for at least 20 seconds. Next, remove the flip-off caps from both the peptide and diluent vials. Using a fresh 70% isopropyl alcohol swab, clean each rubber stopper with firm, circular motions for 15 seconds. Let the stoppers air dry for 15–30 seconds - never blow on or wipe them dry, as this can introduce contaminants.
Always re-sanitize the stoppers before each needle insertion, even if only a short time has passed.
"Sterilize before every needle insertion - not just the first time" – PeptideReconstitute.com Editorial Team
Adding the Diluent
Once your vials are sterile, proceed with adding the diluent. Start by drawing air into the syringe equal to the volume of diluent you’ll need. Inject this air into the diluent vial to equalize the pressure. Slowly draw the required amount of diluent into the syringe.
When injecting the diluent into the peptide vial, insert the needle at an angle and aim for the inside of the glass wall, not directly at the powder. Depress the plunger gradually over 30–60 seconds, letting the liquid trickle down the glass and gently flow over the powder. Spraying the liquid directly onto the powder can lead to mechanical denaturation, reducing potency by 10–30%.
Mixing Without Damaging the Peptide
Avoid shaking the vial - this can create foam and shear forces that damage the peptide chains. Instead, gently swirl the vial in a circular motion, tilting it at a 30–45° angle to ensure the solvent contacts all particles. If the peptide doesn’t dissolve completely after 5 minutes, refrigerate the vial for 15–30 minutes. Once the solution appears fully dissolved, confirm that no particles remain.
Checking for Complete Dissolution
The time it takes for peptides to dissolve depends on their size. Smaller peptides, like BPC-157, usually dissolve within 30 seconds to 2 minutes. Medium-sized peptides, such as Semaglutide, may take 1–5 minutes, while larger proteins like HGH might require 5–15 minutes.
The final solution should be crystal clear and colorless, with no visible particles, foam, or cloudiness. If the solution remains cloudy, discolored, or contains debris after 30 minutes, it should be discarded. Once fully dissolved, store the vial immediately in the refrigerator at 36–46°F (2–8°C), avoiding the door to minimize temperature fluctuations.
Calculating Your Dosage
Once your peptide is dissolved, the next step is determining the liquid volume for each dose. Your solution's concentration dictates how many syringe units correspond to your desired dose. Peptides are measured in milligrams (mg) but are typically administered in micrograms (mcg), where 1 mg equals 1,000 mcg.
Insulin syringes (U-100) are commonly used for this process. These syringes hold 1 mL and are divided into 100 units, meaning each unit equals 0.01 mL. The concentration of your peptide per syringe unit depends on how much bacteriostatic water you used during reconstitution. Use the formula and table below to ensure accurate calculations.
"The amount of solvent you add determines the concentration of your solution, which determines how many units on your syringe equal your desired dose. Get this wrong, and every subsequent measurement will be off." – PeptideReconstitute.com Editorial Team
Dosage Calculation Formula
To calculate your solution's concentration, divide the total peptide amount by the volume of water added. Then, convert the result to micrograms. For example, if you reconstitute 5 mg of peptide with 2 mL of water, your concentration will be 2.5 mg/mL or 2,500 mcg/mL. To find the mcg per syringe unit, divide the concentration by 100: 2,500 ÷ 100 = 25 mcg per unit. Now, to determine how many units to draw for your dose, divide your target dose by the mcg per unit. For a 250 mcg dose: 250 ÷ 25 = 10 units.
Stick to doses between 5 and 50 units for better accuracy. Doses below 5 units are hard to measure, while doses above 50 units require unnecessarily large injection volumes. A 10% error in the amount of solvent used during reconstitution translates to a 10% error in dosing for the entire protocol. To avoid confusion, label your vial immediately after mixing, noting the final concentration (e.g., 2.5 mg/mL) and the date.
| Goal | Formula |
|---|---|
| Find Concentration (mcg/mL) | (Peptide mg × 1,000) ÷ BAC Water mL |
| Find mcg per Syringe Unit | Concentration (mcg/mL) ÷ 100 |
| Find Units to Draw for Dose | (Desired Dose mcg ÷ Concentration mcg/mL) × 100 |
| Find Required Water Volume | (Peptide mg × 1,000) ÷ Desired Concentration (mcg/mL) |
Dosage Reference Table
Here’s a quick reference for common reconstitution scenarios:
| Peptide Amount (Vial Size) | Bacteriostatic Water Added | Concentration (mcg/mL) | mcg per 10 Units (0.1 mL) | mcg per 1 Unit (0.01 mL) |
|---|---|---|---|---|
| 2 mg | 1 mL | 2,000 mcg/mL | 200 mcg | 20 mcg |
| 5 mg | 1 mL | 5,000 mcg/mL | 500 mcg | 50 mcg |
| 5 mg | 2 mL | 2,500 mcg/mL | 250 mcg | 25 mcg |
| 10 mg | 2 mL | 5,000 mcg/mL | 500 mcg | 50 mcg |
| 10 mg | 5 mL | 2,000 mcg/mL | 200 mcg | 20 mcg |
A common starting point is reconstituting 5 mg of peptide with 2 mL of bacteriostatic water. This results in a concentration of 250 mcg per 10 units (0.1 mL), making calculations straightforward. For smaller doses (under 100 mcg), consider using 2–3 mL of water to avoid dealing with fractions of a syringe unit. Remember, increasing the water volume doesn’t change the total peptide amount in the vial - it simply reduces the concentration, making precise measurements for small doses easier.
Storing Reconstituted Peptides
Proper Refrigeration
Once your peptides are reconstituted, proper storage is critical to preserving their effectiveness. The best practice is to refrigerate them immediately. Aim for a temperature range of 36–46°F (2–8°C) - a standard setting for most household refrigerators. Why is this so important? Because for every 10°C increase in temperature, the degradation rate of peptides doubles. Keeping them cold is non-negotiable.
Store the vials upright on the back middle shelf of your fridge. This spot tends to have the most stable temperature and avoids unnecessary contact with the stopper, which could compromise the solution. If your peptides are sensitive to light, protect them by wrapping the vial in aluminum foil or storing it in the original box.
Never freeze reconstituted peptides. Freezing can break peptide bonds, rendering them biologically inactive. While lyophilized (freeze-dried) peptides can be safely stored in the freezer for extended periods, introducing bacteriostatic water changes the game. Freezing the solution damages its integrity. When you need to use your peptide, take the vial out of the fridge for no more than five minutes to draw your dose, then return it promptly.
How Long Peptides Stay Stable
When stored properly with bacteriostatic water, most reconstituted peptides remain stable for 28 to 30 days. The benzyl alcohol in bacteriostatic water helps inhibit bacterial growth during this period. However, if you used sterile water instead (which lacks preservatives), the storage window shrinks drastically to just 24–48 hours.
During the first 28 days, potency typically stays at 95–100%. By day 60, this can drop to about 80–85%. The stability of peptides varies depending on their structure. For example, NAD+ solutions last only 7–14 days, while cyclic peptides like Melanotan II can retain potency for 30–40 days due to their more robust structure. This underscores the importance of proper handling and storage to maintain dosing accuracy. Following strict peptide harm reduction protocols is essential when conducting research outside of clinical settings.
Before each use, examine your solution for signs of degradation or contamination. Here's what to watch for:
| Sign | Meaning | Required Action |
|---|---|---|
| Cloudy solution | Contamination or aggregation | Discard immediately |
| Visible particles/floaters | Precipitation or contamination | Discard immediately |
| Color change (Yellow/Brown) | Oxidative degradation | Discard immediately |
| Unusual odor | Bacterial growth | Discard immediately |
Common Mistakes and How to Fix Them
Avoiding mistakes is just as important as using the right techniques when handling peptides. Missteps can lead to reduced peptide effectiveness and skewed research results.
Preventing Contamination
Contamination is one of the biggest threats to peptide stability. Every time a needle pierces a vial, there's a chance for contamination to occur. To minimize this risk, always use a new, sterile syringe for each step, whether you're drawing or reconstituting. If you're working with multi-dose vials, opt for bacteriostatic water - it contains 0.9% benzyl alcohol, which can keep bacterial growth at bay for up to 28 days. On the other hand, sterile water without preservatives should only be used if the vial will be fully consumed within 24–48 hours. If your solution appears cloudy and contamination isn't the issue, there are other steps to try.
Fixing Cloudy or Clumped Solutions
When contamination has been ruled out but your peptide solution remains cloudy, it doesn't automatically mean the peptide is unusable. Sometimes, the solution just needs more time to dissolve. Larger peptides, like HGH or IGF-1, may take 5–15 minutes to fully dissolve, while smaller ones, such as BPC-157, typically clear up in 30 seconds to 2 minutes. If gentle swirling doesn't resolve the cloudiness within five minutes, refrigerate the vial for 15–30 minutes. However, if the solution is still cloudy after 30 minutes - or shows signs like a yellow or brown tint, floating particles, or an unusual odor - it's best to discard it. These are clear indicators of contamination or degradation. Also, keep in mind that improper mixing techniques can mechanically denature peptides, reducing their effectiveness by 10–30%.
Conclusion
The steps above are key to ensuring safe and effective peptide reconstitution. Precision, sterility, and patience are your best allies in this process. For instance, using 70% isopropyl alcohol to clean the vial top and storing your solution at 36–46°F (2–8°C) are non-negotiable. Techniques like aiming the diluent at the vial wall, gently swirling instead of shaking, and giving the peptide enough time to dissolve - about 30 seconds for BPC-157 or up to 15 minutes for HGH - help protect those fragile peptide bonds.
By using proper techniques, you can avoid potency losses of 10–30% caused by mechanical damage. Refrigeration also plays a big role, extending peptide stability to 28 days with bacteriostatic water, compared to just 24–48 hours with sterile water. Measuring the solvent volume accurately ensures the right concentration, which is crucial for precise dosing during your protocol.
One important rule: never freeze reconstituted peptides. Ice crystals can harm their molecular structure. Following these guidelines helps maintain peptide integrity, ensuring both potency and the reliability of your research.
FAQs
How much bacteriostatic water should I add?
When reconstituting peptides, the volume of bacteriostatic water used usually falls between 1 mL and 10 mL, depending on the concentration you need. For most research applications, 1 mL or 2 mL are common choices. Always refer to the instructions provided with your specific peptide to ensure proper mixing and to achieve the accurate dosage.
What’s the easiest way to calculate syringe units per dose?
To figure out syringe units per dose, start by identifying the concentration of your reconstituted peptide, typically measured in mg/mL. Once you know this, convert the dose you want into milliliters and align it with the syringe's markings.
For instance, if you're working with a 1 mg/mL solution and need a 0.1 mg dose, that translates to 0.1 mL. On a standard insulin syringe, this equals 10 units. This method helps ensure precise dosing and easy administration.
Can I travel with reconstituted peptides without ruining them?
When it comes to traveling with reconstituted peptides, it’s best to avoid it. These peptides need to be kept at a temperature of 2–8°C in a refrigerator and used within 28 days to ensure they remain stable. Exposing them to room temperature or improper storage conditions during travel can lead to degradation.
To prevent this, it’s smarter to transport them in their lyophilized (dry) form. Once you reach your destination, you can reconstitute them and store them correctly to maintain their effectiveness.