Are Peptides Safe? Common Questions Answered

Are Peptides Safe? Common Questions Answered
Peptides are small chains of amino acids used in the body for various functions, such as regulating hormones and aiding in digestion. Their popularity has grown in health and wellness spaces for benefits like weight management, muscle growth, and skin repair. However, safety depends on factors like the peptide's quality, regulatory approval, and individual health conditions. Here's what you need to know:
- FDA-approved peptides undergo rigorous testing and are generally safer than unregulated products.
- Unregulated peptides often lack purity, with some containing impurities or incorrect dosages.
- Common risks include injection-site reactions, nausea, and, in some cases, more severe issues like pancreatitis or hormonal imbalances.
- Certain groups, such as pregnant women, people with cancer, and those with thyroid or kidney issues, should avoid peptides.
- Safe use involves sourcing from trusted, regulated suppliers, medical supervision, and following proper dosing protocols.
Peptides can be effective when used responsibly, but unregulated products and improper use pose serious risks. Always consult a licensed healthcare provider before starting peptide therapy.
Are Peptides Safe? The Clinical Reality
sbb-itb-7fad836
What Makes Peptides Safe or Unsafe?
The safety of peptides hinges on several key factors, including their quality, regulatory status, and the health profile of the individual using them. Let’s break down how these elements influence peptide safety.
Regulated vs. Unregulated Peptides
The origin of a peptide plays a major role in determining its safety. Pharmaceutical-grade peptides, produced under Good Manufacturing Practice (GMP) standards, undergo rigorous quality control at every stage - from raw materials to sterility testing. In contrast, research-grade peptides often come with warnings like "not for human consumption" and lack the same stringent oversight.
Pharmaceutical-grade peptides typically achieve purity levels of 98% or higher, verified through advanced techniques like HPLC (High-Performance Liquid Chromatography) and MS (Mass Spectrometry). On the other hand, unregulated peptides have been found to contain a fraction of their labeled concentration, with published testing data showing some products delivering as little as 7–14% of the intended compound. These products may also include harmful contaminants like truncated sequences, residual synthesis chemicals, or bacterial endotoxins.
"Purity and contamination risks from non‐GMP sources may pose greater danger than the peptide itself." – Peptide Effect
To ensure safety, a Certificate of Analysis (CoA) from an independent, ISO-accredited lab is essential. This document confirms the peptide’s purity, molecular identity, and acceptable levels of endotoxins (typically below 0.25 EU/mL). In-house vendor reports don’t provide the same level of reliability, and unusually low prices can often signal poor quality.
FDA Regulation and Peptide Safety
Regulatory oversight is another critical factor in peptide safety. Peptides approved by the FDA go through extensive testing in Phase 1–3 clinical trials, involving thousands of participants, and remain under continuous monitoring after approval. This process ensures a robust safety profile. By contrast, peptides labeled as "Research Use Only" (RUO) lack human safety data and often rely solely on animal studies, which don’t always predict human outcomes.
On September 29, 2023, the FDA placed BPC-157 on its list of bulk drug substances that present demonstrable difficulties for compounding, prohibiting its use in both 503A and 503B compounding pharmacies. Between 2024 and 2026, broader enforcement against vendors selling research peptides for human use intensified further. As a result, many providers have shifted to sourcing peptides from 503B outsourcing facilities, which comply with stricter CGMP standards and undergo regular FDA inspections.
How Personal Health Affects Peptide Safety
Even when peptides meet the highest quality and regulatory standards, their safety can vary depending on an individual’s health profile. For instance, growth-promoting peptides like Ipamorelin or CJC-1295 may pose risks for individuals with active or recent cancer due to concerns about stimulating tumor growth. Similarly, GLP-1 agonists include boxed warnings for those with a family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN2).
Organ health also plays a role in peptide safety. Impaired kidney or liver function can affect how peptides are metabolized and cleared from the body. In older adults, declining kidney function might prolong a peptide’s half-life, leading to unintended accumulation. For patients with advanced kidney issues (Stage 3 or higher), the risk of adverse effects increases significantly. Additionally, peptides like GLP-1 agonists can slow gastric emptying by 30–50%, which may delay the absorption of medications such as birth control or blood thinners.
Before starting peptide therapy, it’s essential to perform baseline tests and disclose all medications. Common tests include IGF-1, HbA1c, a complete metabolic panel, and thyroid function assessments, which help identify potential risks and ensure safe usage.
Side Effects of Peptides
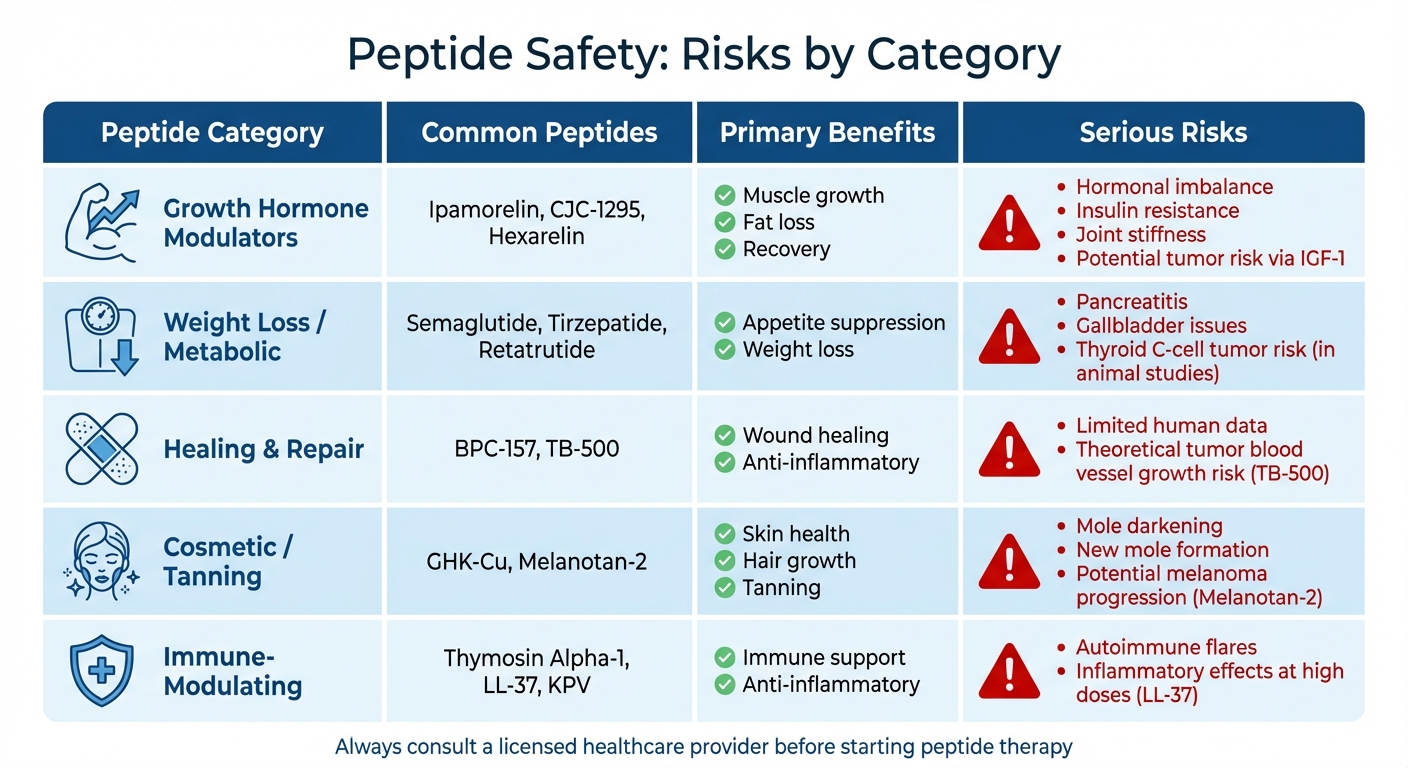
Peptide Safety Risks by Category: Benefits and Serious Side Effects Comparison
Peptides, like any therapeutic option, come with the potential for side effects. These can range from minor inconveniences to more serious health risks. Knowing what to expect can help you make informed decisions and recognize when medical attention might be necessary.
Common Mild Side Effects
Most people experience only mild and temporary side effects. For example, injection-site reactions such as redness, swelling, itching, or pain are common but usually resolve within a few hours to a couple of days. Rotating the injection sites - like switching between the abdomen, thigh, and upper arm - can help reduce irritation and avoid issues like lipodystrophy.
Peptides targeting metabolism may cause stomach-related side effects like nausea or diarrhea, especially during dose adjustments. For instance, clinical trials showed that about 44% of users taking a full 2.4 mg dose of semaglutide experienced nausea, while tirzepatide users reported nausea (31%) and diarrhea (23%).
Growth hormone secretagogues, including Ipamorelin and CJC-1295, may cause sensations like mild hunger or tingling in the hands and feet. Acute flushing sensations post-injection typically resolve within 30–60 minutes, but GH-mediated paresthesia can persist for days to weeks before resolving as the body adapts. Other reported side effects include water retention, headaches, and fatigue, which are usually short-lived. Melanotan-2 commonly causes nausea; clinical studies report rates of approximately 13% at standard doses, though subjective user reports suggest it may be higher in practice.
"If anything feels different or anything happens in the 24 to 72 hours after injection, that could be a side effect. But it's the Wild West because you don't know." – Lauren A. Fine, MD, FAAD, Board-certified dermatologist
While these mild effects are often manageable, some peptides come with more serious risks that demand closer attention.
Serious Risks by Peptide Type
Certain peptides pose significant risks that should not be overlooked. Individual health conditions and the quality of the peptides play a major role in determining these risks.
Weight loss peptides like semaglutide and tirzepatide have been linked to conditions like acute pancreatitis and gallbladder disorders. In clinical studies, gallbladder issues occurred in 2.6% of semaglutide users versus 1.2% in the placebo group. Additionally, these peptides carry boxed warnings about thyroid C-cell tumors based on rodent studies. The triple-agonist retatrutide has also been associated with dysesthesia (an abnormal nerve-related sensation) in approximately 21% of participants at the 12 mg dose in Phase 3 trial data (TRIUMPH-4, 2025).
Growth-promoting peptides may raise IGF-1 levels into ranges associated with elevated cancer risk — often without any noticeable symptoms — underscoring the importance of regular monitoring. They may also cause insulin resistance, joint stiffness, and carpal tunnel-like symptoms. To stay safe, users of these peptides should monitor IGF-1 levels every 8 to 12 weeks.
Cosmetic peptides like Melanotan-2 have documented risks, including the darkening of existing moles, the formation of new moles, and a potential link to melanoma progression. Similarly, healing peptides such as TB-500 raise concerns about promoting tumor blood vessel growth (angiogenesis), though this hasn’t been proven in human studies. Immune-modulating peptides like Thymosin Alpha-1 or LL-37 may trigger autoimmune flares or interfere with immunosuppressive treatments.
| Peptide Category | Common Peptides | Primary Benefits | Serious Risks |
|---|---|---|---|
| Growth Hormone Modulators | Ipamorelin, CJC-1295, Hexarelin | Muscle growth, fat loss, recovery | Hormonal imbalance, insulin resistance, joint stiffness, potential tumor risk via IGF-1 |
| Weight Loss / Metabolic | Semaglutide, Tirzepatide, Retatrutide | Appetite suppression, weight loss | Pancreatitis, gallbladder issues, thyroid C-cell tumor risk (in animal studies) |
| Healing & Repair | BPC-157, TB-500 | Wound healing, anti-inflammatory | Limited human data; theoretical tumor blood vessel growth risk (TB-500) |
| Cosmetic / Tanning | GHK-Cu, Melanotan-2 | Skin health, hair growth, tanning | Mole darkening, new mole formation, potential melanoma progression (Melanotan-2) |
| Immune-Modulating | Thymosin Alpha-1, LL-37, KPV | Immune support, anti-inflammatory | Autoimmune flares, inflammatory effects at high doses (LL-37) |
To reduce risks, peptides should always be reconstituted with bacteriostatic water (0.9% benzyl alcohol) to prevent bacterial growth. Proper handling and high-quality sourcing are critical to avoid contamination.
"The absence of reported side effects in compounds that have not undergone formal safety evaluation should not be interpreted as evidence of safety." – Peptide Effect
Who Should Avoid Peptide Therapy?
Peptide therapy isn't suitable for everyone. While it may provide certain advantages, some people face increased risks due to specific health conditions or circumstances. Knowing who should steer clear of peptide therapy is essential to avoid serious complications.
People Who Should Not Use Peptides
Pregnant and breastfeeding women are advised against peptide therapy. These groups are typically excluded from clinical trials, leaving a gap in safety data regarding potential effects on fetal development or nursing infants. Research on animals has shown that GLP-1 receptor agonists can result in skeletal abnormalities and reduced fetal growth at doses similar to those used in humans. If you are planning to become pregnant, it's recommended to stop peptide therapy at least two months prior to conception.
"Preclinical animal studies have shown teratogenicity at doses similar to those used in humans. Offspring of pregnant animals exposed to GLP1RAs have shown skeletal abnormalities and reductions in fetal weight and growth." – Jonathan S. Zipursky, MD PhD, University of Toronto
Individuals with active cancer or a history of malignancy should avoid growth-promoting peptides such as Ipamorelin, CJC-1295, TB-500, or Follistatin-344. These compounds could potentially accelerate tumor growth by promoting cell proliferation and angiogenesis. Consulting with an oncologist is critical if you have or had cancer and are considering peptide therapy.
People with thyroid-related risks, particularly those with a personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN2), should avoid GLP-1 agonists like semaglutide and tirzepatide. These medications carry warnings about thyroid C-cell tumors.
Those with kidney or liver disease face heightened risks. Impaired organ function can lead to toxic accumulation of peptides, especially in individuals with stage 3 or higher chronic kidney disease or significant liver impairment.
Individuals with autoimmune disorders, such as lupus or rheumatoid arthritis, need to be cautious with immune-modulating peptides like Thymosin Alpha-1 or LL-37. These compounds might trigger disease flares or interfere with treatments designed to suppress the immune system.
People with uncontrolled diabetes should be mindful of growth hormone-stimulating peptides, as these can reduce insulin sensitivity and disrupt glucose control.
Cardiovascular patients, including those with heart failure, high blood pressure, or a history of blood clots, should avoid peptides that affect fluid balance or vascular tone, as they may worsen these conditions.
Minors under 18 should not use peptide therapies. These compounds can interfere with normal hormonal development during puberty.
"The decision to use a peptide should account for diagnosis, comorbidities, other medications, pregnancy status, cancer history, organ function, and intended treatment goals. The internet routinely skips that workup." – Dr. Jossy Onwude, MD
Summary of Risks by Condition
| Condition | Peptides to Avoid | Why It's Risky |
|---|---|---|
| Active Cancer | GH Secretagogues, TB-500, Follistatin-344 | May promote tumor growth and angiogenesis |
| Thyroid History (MTC/MEN2) | Semaglutide, Tirzepatide, Retatrutide | Thyroid tumor risk |
| Kidney Disease (Stage 3+) | BPC-157, Semaglutide, Tirzepatide | Risk of toxic accumulation due to impaired clearance |
| Autoimmune Disease | Thymosin Alpha-1, LL-37 | May trigger immune flares or interfere with immunosuppressants |
| History of Melanoma | Melanotan-2 | Could stimulate melanocytes and promote lesion progression |
How to Use Peptides Safely
To minimize risks and side effects, it’s crucial to focus on proper sourcing, medical oversight, and adhering to dosage instructions. Each step plays a vital role in ensuring peptides are used responsibly.
How to Source Peptides Safely
Always obtain peptides from trusted and regulated sources. For FDA-approved peptides, use a licensed pharmacy. Currently, there are over 80 FDA-approved peptide drugs available. If compounded peptides are necessary, ensure they come from FDA-registered 503B outsourcing facilities. These facilities follow strict Current Good Manufacturing Practice (cGMP) guidelines and undergo regular FDA inspections.
Steer clear of products labeled as "for research purposes only" or "not for human consumption." These products lack regulatory oversight, leaving their purity, sterility, and identity unverified. A 2024 study published in JMIR (Ashraf et al.) found that compounded semaglutide products purchased from unregulated online sources contained only 7.7–14.4% of the labeled active compound — well below therapeutic levels.
Before using any peptide, request a batch-specific Certificate of Analysis (COA) from an ISO-accredited lab. A reliable COA should confirm the following:
- HPLC purity of at least 98%
- Molecular weight and identity verified via mass spectrometry
- Endotoxin levels meeting USP ⟨85⟩ limits (0.25 EU/mL for Water for Injection; drug-specific limits vary and are calculated per USP formula)
- Documentation of sterility testing
"Peptide quality is not something a buyer can infer from a pretty website, a certificate screenshot, or an influencer endorsement." – Dr. Jossy Onwude, MD
Pay attention to shipping conditions, too. Peptides are delicate and can degrade if not stored and shipped correctly. Vendors should use cold-chain shipping to maintain stability. Be cautious of sellers offering prices far below market rates - quality peptide synthesis is costly. Once you’ve secured safe products, medical supervision becomes the next critical step.
Why Medical Supervision Matters
Healthcare providers play a key role in ensuring peptides are used safely. They can screen for contraindications and adjust dosages to minimize side effects. For instance, GLP-1 agonists often require gradual dose increases, as up to 44% of users report gastrointestinal issues.
Medical oversight also helps manage potential drug interactions. GLP-1 agonists, for example, slow gastric emptying by 30% to 50%, which can delay the absorption of medications like oral contraceptives, thyroid treatments, and blood thinners. Providers may also monitor specific markers, such as IGF-1 for growth hormone secretagogues or HbA1c for metabolic peptides, at 8- to 12-week intervals to catch issues early.
Physician-supervised programs ensure access to pharmaceutical-grade peptides sourced from FDA-registered facilities, reducing the risks tied to unregulated products. For example, telehealth programs offering GLP-1 weight loss treatments typically start at around $199 per month.
"Interest is not evidence, and access is not the same as safety." – Dr. Jossy Onwude, MD
With professional guidance, you can proceed to careful dosing practices.
Monitoring and Following Dosage Instructions
Start with the lowest effective dose and introduce only one peptide at a time. Gradual titration is key. For example, increasing Tirzepatide doses every 4 weeks instead of taking a higher dose right away allows your body to adjust and reduces side effects.
Keeping a daily symptom diary for 2–4 weeks can also help. Record the dose, time, injection site, and any changes you notice. This practice makes it easier for you and your healthcare provider to spot patterns early. Rotate subcutaneous injection sites (abdomen, thigh, or upper arm) to avoid issues like localized fat loss or hardening (lipodystrophy).
When reconstituting multi-use vials, use bacteriostatic water (0.9% benzyl alcohol) to prevent bacterial growth for up to 28 days. If using sterile water, discard any unused portion within 24 hours to avoid contamination.
Never exceed prescribed dosages or use unapproved products. The FDA has reported cases where users made 10-fold dosing errors when measuring from multidose vials. If you experience severe abdominal pain (a potential sign of pancreatitis), difficulty breathing (possible allergic reaction), or symptoms of infection at the injection site (such as fever or pus), seek immediate medical attention.
Legal and Regulatory Rules for Peptides in the U.S.
Understanding the legal framework around peptides is key to ensuring their safe and legitimate use. These rules are designed to protect consumers from counterfeit or unsafe products while guiding their proper application.
FDA Oversight and Consumer Protection
Under the Federal Food, Drug, and Cosmetic (FD&C) Act, substances intended to treat, prevent, or affect the body's structure or function are classified as drugs and fall under FDA regulation. This includes peptides when used for therapeutic purposes.
FDA approvals are specific to certain conditions. For instance, semaglutide is approved for managing type 2 diabetes and obesity but not for purposes like longevity or muscle building. While physicians can legally prescribe medications for "off-label" uses, manufacturers are prohibited from marketing them for such purposes. Interestingly, about 20% of prescription drug use in the U.S. is off-label.
Compounding pharmacies are categorized as 503A (state-regulated, patient-specific) or 503B (FDA-inspected and compliant with cGMP standards). As of mid-2025, the U.S. has approximately 90–100 FDA-registered 503B facilities, compared to thousands of 503A pharmacies. The FDA maintains a "Bulk Drug Substance List" for compounding that prohibits certain substances labeled Category 2 due to safety or efficacy concerns. On September 29, 2023, the FDA added approximately 19–20 bulk drug substances — including BPC-157, CJC-1295, and Ipamorelin — to its Category 2 list, prohibiting their use in compounding pharmacies.
Peptides labeled as "Research Use Only" (RUO) are strictly for laboratory testing. Using these for personal use violates their classification and offers no consumer protections. Studies have revealed issues with RUO peptides, including inconsistent concentrations, and independent testing has identified impurity and potency issues across a substantial proportion of gray-market research-grade peptides.
The FDA enforces compliance through warning letters, import alerts, and even criminal prosecution via the Department of Justice. For instance, multiple research peptide vendors have received FDA enforcement actions for marketing peptides like BPC-157 and TB-500 with therapeutic claims such as "healing" and "recovery," which constitutes misbranding under the FD&C Act. Following these regulations is just as important as adhering to medical guidelines for safe peptide use.
Legal Risks of Unregulated Peptides
Using unregulated peptides not only poses health risks but also carries legal consequences. For example, in 2020, Tailor Made Compounding LLC (Nicholasville, KY) pleaded guilty to distributing unapproved drugs, including BPC-157, across state lines, resulting in a $1.79 million forfeiture — an early indicator of federal willingness to criminally prosecute compounding violations.
Since 2025, state attorneys general have increasingly used consumer protection laws to target sellers of unapproved peptides, even without federal FDA action. Multiple state attorneys general have taken action to address the risks posed by unapproved online peptide sales, with enforcement activity increasing significantly through 2025 and into 2026 [15].
Some states have gone beyond federal enforcement — for example, Connecticut's Attorney General issued a May 2025 consumer protection warning targeting sellers of unapproved compounded semaglutide under state unfair trade practices law.
To ensure compliance, verify whether your pharmacy is registered as 503A or 503B through state pharmacy boards. Before using a compounded peptide, check its status on the FDA's bulk drug substances list. Be cautious of vendors that don’t require prescriptions, ship internationally, or provide dosing instructions for products labeled "not for human consumption". Athletes should also cross-check peptides against the WADA Prohibited List, as many legal research peptides are banned in competitive sports.
In February 2025, the FDA removed branded semaglutide products (Ozempic, Wegovy) from its drug shortage list, significantly restricting (though not entirely eliminating) their compounding in 503A pharmacies — compounding may still occur in limited circumstances involving documented patient-specific clinical need. This shift highlights how quickly regulations can change, underscoring the importance of staying informed. These rules not only aim to prevent unsafe products but also shield consumers from potential legal issues.
Conclusion
Peptide safety is a complex topic. As Dr. Jossy Onwude, MD, explains:
"Peptide is not a safety category. Safety depends on the specific molecule, the indication, the dose, the formulation, the source, and the quality of supervision."
With over 80 FDA-approved peptide drugs available, the key to safety lies in proper manufacturing and strict monitoring. The real risks often stem from unregulated products.
Your safety hinges on three critical factors: sourcing peptides from regulated facilities that provide third-party Certificates of Analysis (ensuring HPLC purity above 98%), collaborating with licensed healthcare providers who monitor essential biomarkers every 8–12 weeks, and adhering to gradual, evidence-based dosing protocols. These steps can mean the difference between achieving therapeutic benefits and encountering serious risks.
Many naturally occurring peptides break down into amino acids rather than toxic metabolites, which can reduce certain long-term safety concerns — though modern therapeutic peptides are often chemically modified specifically to resist this breakdown, and their metabolic profiles require individual assessment. However, this doesn’t eliminate all risks. For example, growth-promoting peptides may raise IGF-1 levels into ranges associated with elevated cancer risk — often without any noticeable symptoms — underscoring the importance of regular monitoring. This is why regulatory updates and medical supervision are so important; they help identify and address these hidden dangers before they escalate.
As highlighted earlier, product quality, medical oversight, and careful dosing are essential for reducing risks. Peptides can be a safe and effective therapy when sourced responsibly, used under professional guidance, and administered following well-researched protocols. Ignoring these precautions, however, can turn a promising treatment into a serious hazard.